Implantable Bioresorbable Scaffold with FCS: The Future of Hematopoietic Therapy
October 31, 2025
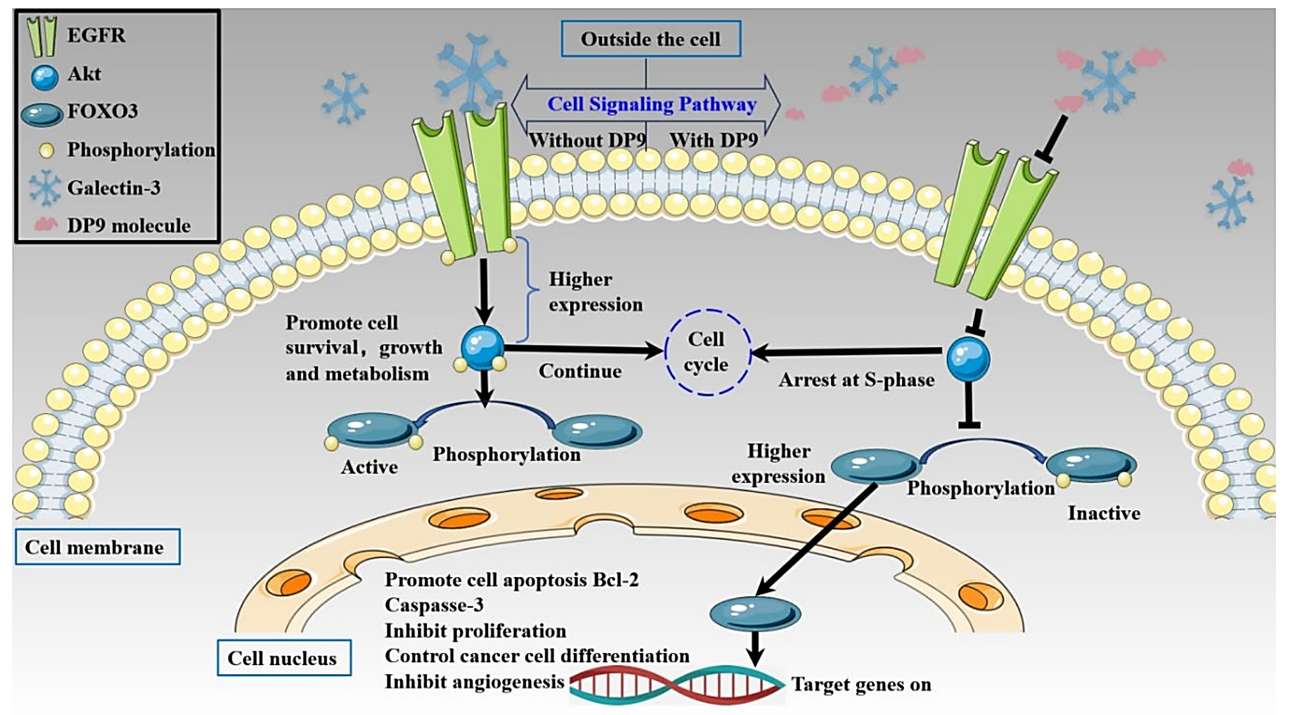
On August 28, 2025, Anisimova et al. published an article in Marine Drugs, titled "Implantable Bioresorbable Scaffold with Fucosylated Chondroitin Sulfate as a Promising Device for Delayed Stimulation of Hematopoiesis". The research validates a novel method for sustained hematopoietic support. The central innovation involves incorporating Fucosylated Chondroitin Sulfate (FCS), a potent glycosaminoglycan derived from marine invertebrates, into a bioresorbable metallic scaffold composed of Fe–Mn–C. The main finding demonstrates that this implanted construct provides the most effective and complex stimulation of hematopoiesis in vivo, leading to rapid and comprehensive recovery of critical blood cell populations and robust regeneration of progenitor cells within the bone marrow.
Research Background
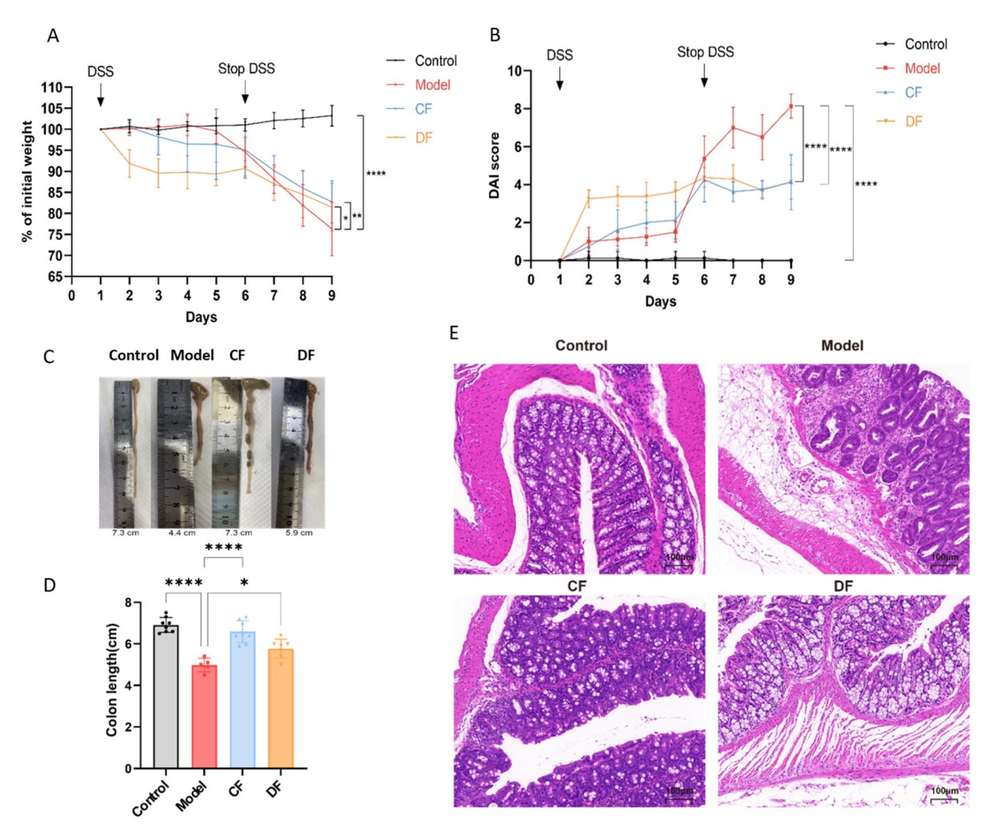
Chemotherapy-induced myelosuppression is a major complication in cancer patients, characterized by neutropenia (the most common), anemia, thrombocytopenia, and even life-threatening pancytopenia. This not only increases infection risk and reduces quality of life but also forces dose reductions or delays in chemotherapy, compromising treatment efficacy. Natural sulfated polysaccharides like FCS—extracted from the sea cucumber Cucumaria japonica—have previously shown hematopoietic-stimulating activity in cyclophosphamide (CPh)-induced myelosuppressive animals. This study aimed to develop an implantable Bioresorbable Scaffold loaded with FCS or recombinant human granulocyte colony-stimulating factor (rG-CSF) for delayed drug release, overcoming the limitations of current therapies, and to evaluate its efficacy in mitigating CPh-induced myelosuppression.
Research Results
- Engineering the Bioresorbable Metal Scaffold
The researchers successfully developed a novel bioresorbable metallic scaffold using Fe–Mn–C and Fe–Mn–Pd alloys. Bioresorbable metals offer a significant advantage over non-resorbable materials, as they dissolve safely in the body over time, eliminating the need for removal surgery. This scaffold was designed not only for structural integrity but also to serve as an efficient reservoir for the therapeutic agent. The use of specific metal alloys was critical, as their controlled degradation profile dictates the drug release kinetics, directly addressing the challenge of rapid clearance seen with traditional methods.
 Fig.1 Scaffold scheme: 1-hollow cylinder made of Fe-Mn-C and Fe-Mn-Pd alloys; 2-mixture of gel and the preparation; 3-outer layer (polyethylene glycol). (Anisimova, et al., 2025)
Fig.1 Scaffold scheme: 1-hollow cylinder made of Fe-Mn-C and Fe-Mn-Pd alloys; 2-mixture of gel and the preparation; 3-outer layer (polyethylene glycol). (Anisimova, et al., 2025)
The therapeutic payload is a marine-derived sulfated polysaccharide: FCS. Isolated from the sea cucumber Cucumaria japonica, FCS is a prime example of the pharmacological power hidden within marine organisms. The study confirmed the effective loading of FCS onto the metallic surface of the scaffold. FCS is known to exert hematopoiesis-stimulating activity, often mimicking the function of natural growth factors. By embedding this glycosaminoglycan into the scaffold, the researchers developed a synergistic system where the biomaterial governs the release profile of the highly active Marine Molecule.
- Sustained Glycan Release Promotes Hematopoietic Recovery
The key to this study's success lies in the in vivo efficacy of the delayed-release mechanism. Using a mouse model of chemotherapy-induced immunosuppression (CPh), the team compared the implantation of the FCS-loaded scaffold against the administration of soluble FCS or rG-CSF. The implanted scaffolds resulted in a significantly more effective and sustained restoration of peripheral blood cell counts, including leukocytes, neutrophils, and lymphocytes. The delayed release of the FCS glycan from the resorbable scaffold provided a prolonged therapeutic window, maximizing bone marrow cell proliferation (Ki-67 labeling) and splenic recovery, which are crucial markers for successful hematopoietic regeneration.
 Fig.2 Morphology of the spleen of mice with CPh-induced immunosuppression after implantation. (Anisimova, et al., 2025)
Fig.2 Morphology of the spleen of mice with CPh-induced immunosuppression after implantation. (Anisimova, et al., 2025)
Conclusion
This research convincingly concludes that combining an active Marine Glycan, FCS, with a bioresorbable metallic scaffold is a highly effective strategy for the delayed and sustained stimulation of hematopoiesis. The innovative implantable device overcomes the pharmacokinetic challenges of administering soluble therapeutic agents, providing a prolonged stimulus essential for full bone marrow and immune recovery. This work establishes a pivotal benchmark for future therapeutic interventions, demonstrating the potential of marine glycobiology to provide novel, high-impact solutions for biomaterial-mediated Drug Delivery in critical care and regenerative medicine.
Reference
- Anisimova, N.Y.; et al. Implantable bioresorbable scaffold with fucosylated chondroitin sulfate as a promising device for delayed stimulation of hematopoiesis. Marine Drugs. 2025, 23(9): 344. (Open Access)

 Fig.1 Scaffold scheme: 1-hollow cylinder made of Fe-Mn-C and Fe-Mn-Pd alloys; 2-mixture of gel and the preparation; 3-outer layer (polyethylene glycol). (Anisimova, et al., 2025)
Fig.1 Scaffold scheme: 1-hollow cylinder made of Fe-Mn-C and Fe-Mn-Pd alloys; 2-mixture of gel and the preparation; 3-outer layer (polyethylene glycol). (Anisimova, et al., 2025) Fig.2 Morphology of the spleen of mice with CPh-induced immunosuppression after implantation. (Anisimova, et al., 2025)
Fig.2 Morphology of the spleen of mice with CPh-induced immunosuppression after implantation. (Anisimova, et al., 2025)