Chitosan-Mediated Expression of Caenorhabditis elegans fat-1 and fat-2 in Sparus aurata
November 3, 2025
On November 13, 2025, researchers led by I. Metón and colleagues published an article in the journal Marine Drugs titled "Chitosan-Mediated Expression of Caenorhabditis elegans fat-1 and fat-2 in Sparus aurata: Short-Term Effects on the Hepatic Fatty Acid Profile, Intermediary Metabolism, and Proinflammatory Factors." The paper investigates the use of chitosan—a primary Marine-derived Polysaccharide—as a non-viral vector to deliver Caenorhabditis elegans genes, fat-1 and fat-2, into the liver of the gilthead seabream (Sparus aurata). This work demonstrates a potent, short-term metabolic reprogramming that significantly alters the fish's hepatic fatty acid profile, enhancing the biosynthesis of essential n-3 long-chain polyunsaturated fatty acids (LC-PUFAs) and downregulating proinflammatory signaling. The use of Chitosan, derived from the shells of marine crustaceans, elegantly integrates a sustainable marine resource into a cutting-edge biotechnological solution, offering a highly promising pathway to enhance the nutritional quality and resilience of farmed fish.
Research Background
Modern aquaculture faces a critical sustainability bottleneck: the global demand for essential n-3 LC-PUFAs, which must primarily be provided in fish feed via finite sources like wild-caught fish oil. As wild fish stocks decline, the industry requires innovative, biotech-driven solutions to enhance the nutritional value of farmed species without relying on unsustainable inputs. This background necessitated the development of non-feed-based metabolic solutions. The utilization of Chitosan, a readily available marine biomass waste product, as the Gene Delivery Vehicle creates a closed-loop marine biotechnology solution, adding significant value and sustainability to the approach.
Research Results
The study systematically evaluated the short-term physiological and molecular impacts of delivering genetic material using a sophisticated, marine-derived delivery system, highlighting the multi-faceted benefits of this glycobiology approach. The experiments were meticulously designed to observe immediate changes in the liver, the central hub of lipid metabolism.
- Glycoconjugate-Enabled Non-Viral Gene Delivery
The foundational innovation centers on the carrier system. The authors successfully utilized chitosan, a deacetylated derivative of chitin, complexed with tripolyphosphate (TPP) to form stable, positively charged nanoparticles. These chitosan-TPP nanoparticles acted as a robust, non-viral vector, encapsulating the negatively charged plasmid DNA encoding the C. elegans genes. Intraperitoneal injection into S. aurata confirmed that this glycoconjugate delivery method effectively transfected hepatocytes, leading to the successful expression of the foreign genes in the liver tissue—a critical step for targeted metabolic engineering in a complex marine organism.
- Engineering Essential n-3 LC-PUFA Synthesis
The primary objective was to improve the fish's capacity to synthesize highly valuable n-3 fatty acids, such as eicosapentaenoic acid (EPA) and docosahexaenoic acid (DHA), which are essential for fish health and human nutrition. The delivered fat-1 gene encodes a n-3 desaturase, converting n-6 fatty acids into beneficial n-3 derivatives, while fat-2 encodes a delta 12 and delta 15 desaturase. Results showed that the co-expression of these genes rapidly and substantially modified the hepatic fatty acid composition. The liver, a central metabolic organ, exhibited a significant increase in the ratio of n-3 to n-6 LC-PUFAs, achieving a healthier lipid profile that typically requires resource-intensive fish oil supplementation.
 Fig.1 Fatty acid methyl ester analysis of liver samples from S. aurata. (Wu, et al., 2025)
Fig.1 Fatty acid methyl ester analysis of liver samples from S. aurata. (Wu, et al., 2025)
- Transcriptional Control and Metabolic Shift
The successful expression of fat-1 and fat-2 triggered a widespread transcriptional response, indicating a potent shift in intermediary metabolism. The study observed a substantial downregulation of key lipogenic transcription factors, including peroxisome proliferator-activated receptor gamma (pparg) and sterol regulatory element-binding protein 1 (srebf1). This molecular signature suggests a strong redirection of hepatic lipid processing, shifting the metabolism away from fat accumulation and storage. Concurrently, there was an upregulation of carbohydrate-responsive element-binding protein (chrebp), highlighting a complex but beneficial metabolic re-engineering that favors beneficial lipid turnover.
- Attenuation of Hepatic Proinflammatory Signaling
A significant finding with direct health implications was the marked reduction in the expression of proinflammatory factors in the fish liver following the gene delivery. This effect is directly attributable to the improved n-3/n-6 fatty acid ratio, as n-3 LC-PUFAs are known precursors for anti-inflammatory eicosanoids. By enhancing the fish's ability to produce these essential Anti-inflammatory Compounds internally, the chitosan-mediated gene delivery offers a route to improve overall systemic resilience and health, which is vital for reducing disease incidence in intensive aquaculture systems.
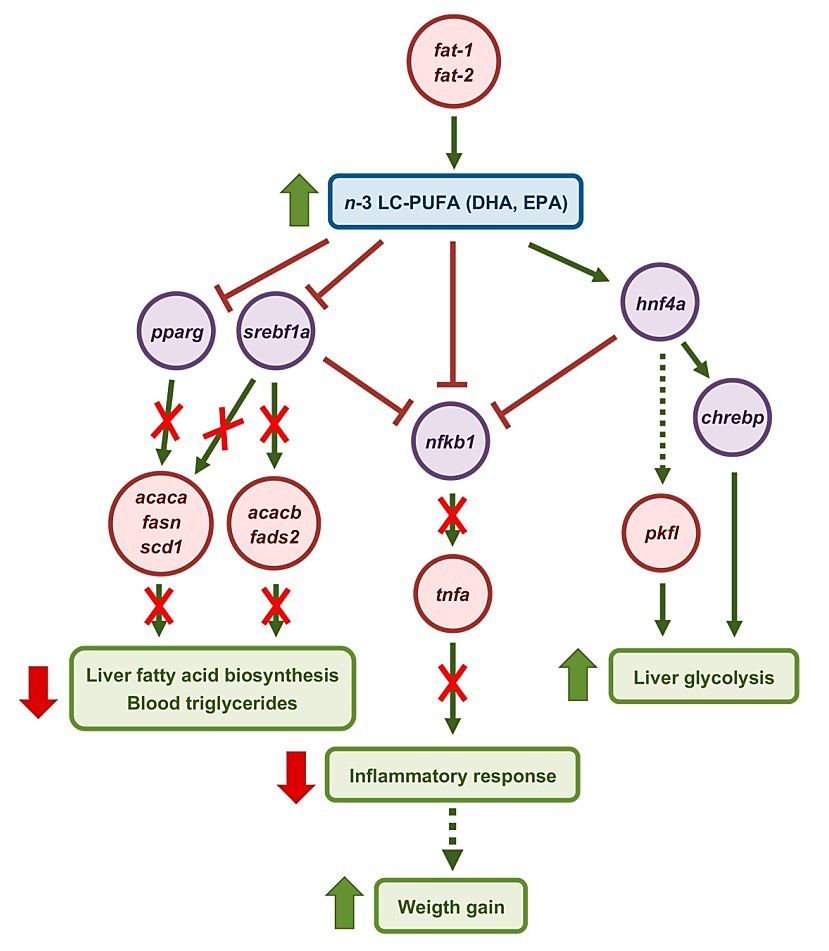 Fig.2 Schematic representation of the short-term molecular events triggered by C. elegans fat-1 and fat-2 in the liver of S. aurata. (Wu, et al., 2025)
Fig.2 Schematic representation of the short-term molecular events triggered by C. elegans fat-1 and fat-2 in the liver of S. aurata. (Wu, et al., 2025)
Conclusion
This research represents a powerful confluence of Marine Glycobiology and aquaculture technology. The effective use of Chitosan Nanoparticles demonstrates a robust, environmentally friendly, and efficient non-viral platform for in vivo gene delivery in marine species. The study's conclusion is compelling: metabolic engineering via this Glycoconjugate Vector can rapidly and effectively enhance the nutritional profile of farmed fish, driving a critical increase in beneficial n-3 fatty acids while simultaneously improving liver health by modulating key metabolic and inflammatory pathways. This dual benefit—nutritional enhancement for consumers and improved health and welfare for the fish—highlights the transformative potential of marine glycans. The transient, yet impactful, gene expression achieved suggests a viable strategy for improving nutritional quality at key points in the fish farming cycle. This innovative application holds immense promise for developing healthier, more nutritious farmed seafood and significantly improving the environmental sustainability of the global aquaculture industry.
Reference
- Wu, Y.; et al. Chitosan-mediated expression of Caenorhabditis elegans fat-1 and fat-2 in Sparus aurata: short-term effects on the hepatic fatty acid profile, intermediary metabolism, and proinflammatory factors. Marine Drugs. 2025, 23(11): 434. (Open Access)

 Fig.1 Fatty acid methyl ester analysis of liver samples from S. aurata. (Wu, et al., 2025)
Fig.1 Fatty acid methyl ester analysis of liver samples from S. aurata. (Wu, et al., 2025) Fig.2 Schematic representation of the short-term molecular events triggered by C. elegans fat-1 and fat-2 in the liver of S. aurata. (Wu, et al., 2025)
Fig.2 Schematic representation of the short-term molecular events triggered by C. elegans fat-1 and fat-2 in the liver of S. aurata. (Wu, et al., 2025)




