Dry eye disease is a prevalent and complex multifactorial condition of the ocular surface. As research increasingly highlights the therapeutic potential of natural compounds, seaweed oligosaccharides have emerged as promising candidates due to their known anti-inflammatory, antioxidant, and moisturizing properties. At CD BioGlyco, we specialize in providing rigorous, high-quality dry eye model efficacy validation services for seaweed oligosaccharides. Our service is designed to precisely evaluate the in vivo and in vitro efficacy of your seaweed oligosaccharide-based compounds, offering critical data to advance your product from discovery to preclinical development. By leveraging our specialized glycobiology expertise and established animal models, we ensure your therapeutic candidates are thoroughly assessed.
Key Technologies
Established In Vivo Dry Eye Models
We utilize several reliable animal models, primarily in rodents (mice and rats) and rabbits, which mimic various forms of dry eye disease, including aqueous-deficient and evaporative types.
High-Resolution Ocular Imaging and Assessment
We employ advanced techniques for objective, non-invasive measurements of the ocular surface, including slit-lamp biomicroscopy, non-invasive tear break-up time (NIBUT) analysis, and meibography.
Glycobiology-Specific Analysis
Our technology includes the capability to analyze the impact of seaweed oligosaccharides on the ocular surface glycocalyx using techniques like Immunofluorescence staining and western blotting.
Dry Eye Model: Validating the Next Generation of Dry Eye Therapeutics
CD BioGlyco offers a specialized, end-to-end dry eye model efficacy validation service for seaweed oligosaccharides, explicitly tailored for compounds derived from marine sources. Our service is designed to move your promising marine-derived therapeutic from the lab bench to proof-of-concept. Key screening and method services directly related to seaweed oligosaccharide validation include:
- Ocular Surface Biomarker Screening
We don't just measure signs; we delve into the molecular mechanism. This involves quantifying the impact of the oligosaccharide on ocular surface mucin expression and glycocalyx integrity. Seaweed oligosaccharides are hypothesized to directly interact with and protect the ocular surface glycocalyx, and our assays are specifically structured to validate this mechanism.
- Anti-Inflammatory Activity Confirmation
A key characteristic of dry eye disease is chronic inflammation. We assess the ability of your oligosaccharide to downregulate key inflammatory pathways by measuring the levels of pro-inflammatory cytokines in the conjunctiva and lacrimal gland tissue.
- Corneal Epithelial Healing Assays
We offer in vitro scratch-wound assays using human corneal epithelial cells (HCEC) to assess the ability of the oligosaccharide to promote cell migration and healing, a critical factor in dry eye disease treatment.
- Toxicology and Tolerability Assessment
Alongside efficacy, we provide initial ocular tolerability testing (e.g., Draize test scores) to ensure the formulation is safe and non-irritating, a crucial prerequisite for ophthalmic products.
Workflow
1. Project Consultation and Design
An in-depth consultation to understand your specific oligosaccharide compound, its source, degree of polymerization, and target mechanism. Then design a customized protocol, selecting the most appropriate dry eye model and dose-response regimen.
2. Dry Eye Model Establishment
We establish the chosen in vivo animal model of dry eye disease. This phase involves strict adherence to ethical guidelines and consistent methodology to generate a stable, reproducible disease state.
3. Oligosaccharide Administration
The seaweed oligosaccharide, formulated into an appropriate delivery vehicle, is administered to the treatment groups according to the predefined dosing schedule. Vehicle-control and positive-control groups are always included for comparative analysis.
4. Endpoint Measurement
Throughout the intervention period, we perform time-point-specific, objective assessments. These include the Schirmer test (STT), tear break-up time (TBUT), and fluorescein and lissamine green staining.
5. Molecular and Histopathological Analysis
Ocular tissues (cornea, conjunctiva, lacrimal gland) and tear samples are collected for advanced analysis. This involves histopathology, immunohistochemistry, etc.
6. Data Analysis and Report
The collected data is subjected to statistical analysis. We deliver a detailed, comprehensive report that includes the raw data, statistical findings, graphical representations of efficacy endpoints, interpretations, etc.
Publication Data
DoI: 10.3389/fmed.2022.830592
Journal: Front. Med
IF: 3.0
Published: 2022
Results: This review systematically summarizes diverse animal models of dry eye disease and evaluates their utility in elucidating dry eye disease pathophysiology and testing therapies. It categorizes models into aqueous-deficient and evaporative types, plus chemical-induced and autoimmune variants. Aqueous-deficient models include surgical lacrimal gland excision, radiation-induced gland damage, neural pathway blockade, and autoimmune models that mimic Sjögren's syndrome with lymphocytic gland infiltration. Evaporative models involve environmental stress, meibomian gland dysfunction, and lid retractor-induced tear evaporation. Chemical-induced models use scopolamine, benzalkonium chloride, or botulinum toxin to trigger dry eye disease. Each model has unique strengths and limitations, highlighting the need for targeted model selection based on research objectives to advance translational dry eye disease research.
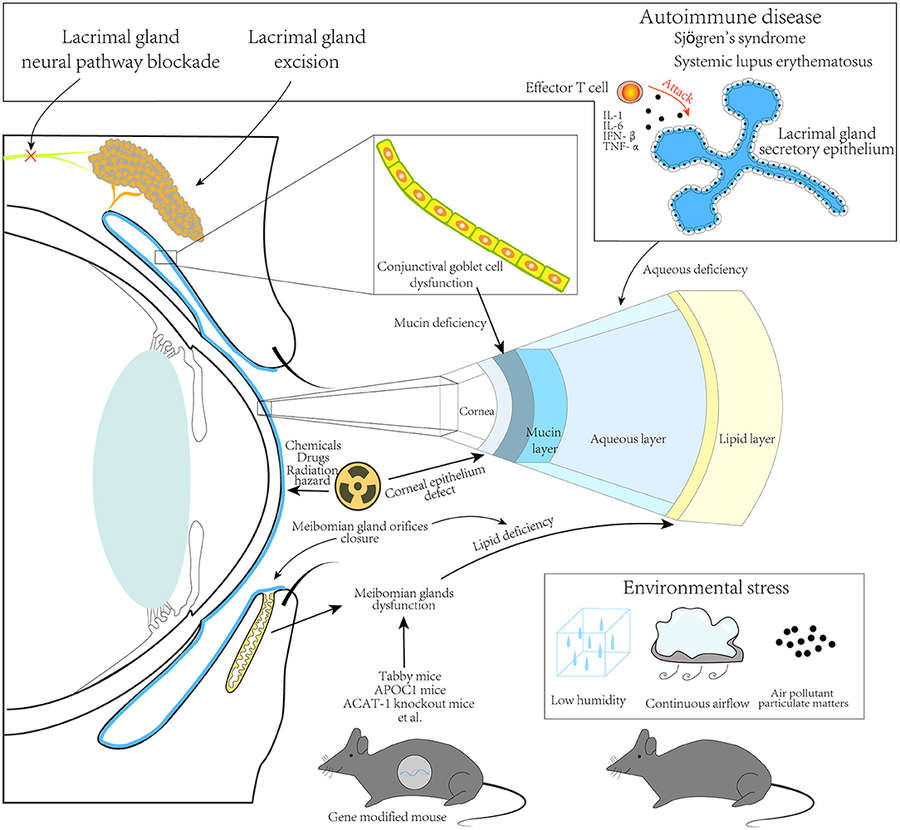 Fig.1 Principles for animal dry eye modeling. (Zhu, et al., 2022)
Fig.1 Principles for animal dry eye modeling. (Zhu, et al., 2022)
Applications
- Drug Development
Validating seaweed oligosaccharides as novel active pharmaceutical ingredients (APIs), particularly those targeting inflammation and tear film stabilization.
- Contact Lens Solutions
Testing the efficacy of oligosaccharides as a component in contact lens rewetting drops, cleaning solutions, or incorporated directly into the contact lens material to improve biocompatibility and moisture retention.
- Ophthalmic Skincare
Validating the moisturizing, anti-inflammatory, and barrier-protecting effects of oligosaccharides for use in eye creams, gels, and other products applied around the eye region that aim to mitigate dryness and irritation.
- Basic Glycobiology Research
Utilizing the validation model to better understand the interaction between specific oligosaccharide structures and the ocular surface glycocalyx, contributing fundamental knowledge to the field of marine-derived glycotherapeutics.
Advantages
- Deep Glycobiology Specialization
We are world-class experts in glycobiology, understanding how saccharides interact with biological systems. This knowledge allows us to design more predictive assays and provide unparalleled mechanistic interpretation.
- Reproducible Animal Models
Our dry eye models are meticulously standardized for low variability, offering high statistical power and reliability for dose-response studies. Our protocols are constantly refined based on the latest scientific literature.
- Customized, Mechanistic-Driven Protocols
We tailor every aspect of the study—from model selection to endpoint analysis—to the unique properties of your seaweed oligosaccharide, focusing on validating its specific mechanism of action (e.g., anti-inflammatory, moisturizing).
- Integrated Efficacy and Safety Assessment
We seamlessly integrate initial ocular toxicity and tolerability testing into the efficacy study, providing a comprehensive risk/benefit profile in a single, efficient study package.
Frequently Asked Questions
Which dry eye model is best for validating my seaweed oligosaccharide?
The optimal model depends on your compound's hypothesized mechanism. For assessing tear-secretion improvement, a scopolamine or excision model may be best. For inflammation, an environmental stress-induced model is often chosen. We will recommend and customize the most predictive model during our initial consultation.
Can you test my oligosaccharide in combination with other ingredients?
Absolutely. We design combination studies to evaluate synergistic or additive effects, which is highly relevant for multi-component ophthalmic formulations (e.g., a combination of an oligosaccharide with a lubricating agent).
Customer Review
"The CD BioGlyco team was instrumental in moving our marine-derived oligosaccharide project forward. Their expertise in glycobiology and the rigor of their environmental dry eye model were exceptional. We received clear, statistically sound data demonstrating a significant improvement in tear stability and corneal health."
- H.L., Director of R&D
"We initially struggled with the formulation for our seaweed compound, but CD BioGlyco's formulation support was top-tier. They successfully prepared a stable ophthalmic solution and executed the mice in vivo efficacy trial flawlessly."
- P.J., Pharmaceutical Company
"What impressed us most was their ability to connect the clinical improvement to the glycobiology. Their analysis confirming the protective effect of our oligosaccharide on the ocular glycocalyx gave us a unique selling point that other CROs couldn't offer."
- A.R., Project Lead
Associated Services
CD BioGlyco is dedicated to advancing the field of glycobiology and supporting the development of innovative, natural therapeutics, particularly for challenging conditions like dry eye disease. Our specialized dry eye model efficacy validation service offers a powerful combination of scientific expertise, rigorous models, and mechanistic insights, ensuring your marine-derived compound achieves its full potential. Please feel free to contact us to discuss your specific requirements.
Reference
- Zhu, J.; et al. Application of animal models in interpreting dry eye disease. Front. Med. 2022, 9: 830592. (Open Access)

 Fig.1 Principles for animal dry eye modeling. (Zhu, et al., 2022)
Fig.1 Principles for animal dry eye modeling. (Zhu, et al., 2022)