Novel Sulfated Oligosaccharide DP9: A Potent Galectin-3 Inhibitor for Pancreatic Cancer Therapy
November 24, 2025
On October 30, 2025, Liu et al. from Guangdong Provincial Key Laboratory of Marine Biotechnology published an article in the journal Marine Drugs titled "Novel Sulfated Oligosaccharide DP9 from Marine Algae, Gracilaria lemaneiformis: A Potent Galectin-3 Inhibitor for Pancreatic Cancer Therapy". The paper investigates the discovery and structural characterization of a novel Sulfated Oligosaccharide, designated DP9, isolated from the common red macroalga Gracilaria lemaneiformis. This study confirms that marine organisms remain an unparalleled source of Bioactive Molecules, with DP9 demonstrating potent and specific inhibitory effects against Galectin-3 (Gal-3), an established oncogenic protein. These findings position DP9 as a leading candidate for developing novel, marine-derived glycotherapeutic strategies for aggressive diseases, particularly pancreatic cancer, where high Gal-3 expression is a major prognostic indicator of poor outcome and chemoresistance.
Research Background
Gal-3 is a β-galactoside-binding lectin ubiquitously expressed in the body, but its overexpression is strongly correlated with malignancy, metastasis, angiogenesis, and resistance to traditional chemotherapy across numerous solid tumors, including pancreatic, breast, and prostate cancer. Gal-3 functions as a critical master regulator of the tumor microenvironment (TME), acting as a structural component of the "glycocode" that governs cell-to-cell and cell-to-matrix interactions. Its pathological actions extend beyond simple cell surface adhesion; it is involved in complex intracellular signaling, capable of inhibiting apoptosis (programmed cell death) and promoting epithelial-mesenchymal transition (EMT), a process essential for metastasis. Inhibiting Gal-3's glycan-binding function is a major therapeutic goal, as this action essentially disarms the protein's ability to drive oncogenesis.
Marine algae, particularly red seaweeds like Gracilaria lemaneiformis, are rich in sulfated galactans (agarans), which are highly similar to the natural ligands recognized by Gal-3. These marine glycans often possess diverse and unique Structural Modifications—such as specific sulfation patterns—that can confer exceptional specificity and potency against lectins compared to simple, non-sulfated human glycans. This makes marine-derived oligosaccharides a prime resource for next-generation Gal-3 inhibitors.
Research Results
- Advanced Isolation and Structural Elucidation of Novel DP9
The initial innovation lies in the successful isolation and precise Structural Analysis of the oligosaccharide. The research employed controlled acid hydrolysis of the high molecular weight polysaccharide from G. lemaneiformis to yield a mixture of oligoagarans. Through techniques like high-performance liquid chromatography (HPLC) coupled with evaporative light scattering detection (ELSD) and fluorophore-assisted carbohydrate electrophoresis (FACE), a single compound with a degree of polymerization (DP) of 9—hence named DP9—was isolated in high purity. Crucially, comprehensive structural analysis using gas chromatography-mass spectrometry (GC-MS), Fourier-transform infrared spectroscopy (FTIR), and extensive nuclear magnetic resonance (NMR) spectroscopy revealed the precise repeating structure and confirmed the presence of unique sulfation patterns. This detailed structural fingerprint confirms DP9 as a novel marine glycan, distinct from previously reported oligoagarans.
- Computational Modeling Confirms Potent Galectin-3 Binding
The paper utilized advanced computational glycobiology tools to predict and validate the interaction between DP9 and Gal-3. Molecular docking simulations demonstrated that DP9 fits snugly into the carbohydrate recognition domain (CRD) of the Gal-3 protein, a process visualized by the researchers using UCSF Chimera to confirm steric compatibility. Simulations indicated that DP9 not only occupies the classic S-subsite, which typically binds the non-reducing β-galactoside unit, but also extends deep into non-classic secondary binding pockets, achieving a significantly higher affinity and more extensive molecular footprint than smaller ligands. Further validation was achieved through rigorous molecular dynamics (MD) simulations, which tracked the stability of the DP9-Gal-3 complex over time. The MD results showed a stable binding affinity, confirming that the specific length and unique sulfation features of DP9 create an optimal molecular interface for targeted inhibition, significantly stabilizing hydrogen bonds and hydrophobic interactions with key residues in the Gal-3 active site.
 Fig.1 Binding conformation of docked DP9 and Gal-3 molecules. (Liu, et al., 2025)
Fig.1 Binding conformation of docked DP9 and Gal-3 molecules. (Liu, et al., 2025)
- In Vitro Efficacy in Pancreatic Cancer Cell Models
Translating the molecular mechanism into functional Biological Activity, the team tested DP9 against BxPC-3 pancreatic cancer cells. Galectin-3 is known to promote tumor progression and metastasis, partially by supporting tumor cell aggregation and tissue invasion. The study demonstrated that DP9 effectively reduced the proliferation and increased the cytotoxicity against these cancer cells in a dose-dependent manner. Furthermore, the oligosaccharide significantly inhibited the ability of BxPC-3 cells to form tube-like structures on Matrigel, an in vitro proxy for angiogenesis and metastatic potential. These results strongly suggest that by blocking Gal-3's activity, DP9 can disrupt critical cellular pathways that drive cancer aggression, offering a promising, non-cytotoxic approach to cancer therapy.
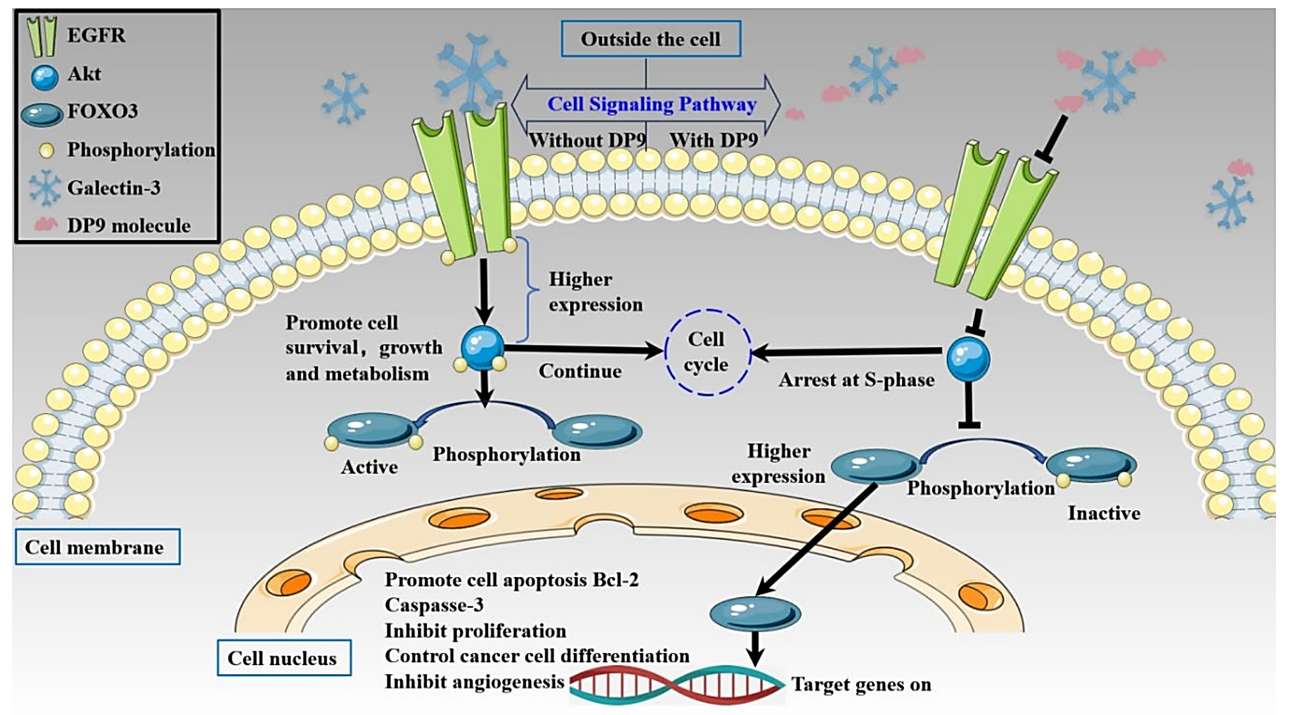 Fig.2 Mechanism of DP9 in the BxPC-3 cell targeting the Gal-3/EGFR/AKT/FOXO3 signaling pathway. (Liu, et al., 2025)
Fig.2 Mechanism of DP9 in the BxPC-3 cell targeting the Gal-3/EGFR/AKT/FOXO3 signaling pathway. (Liu, et al., 2025)
Conclusion
The discovery and comprehensive characterization of DP9 mark a substantial step forward in marine glycotherapeutics. By leveraging the structural diversity of red seaweed components, the research has unveiled an oligosaccharide with validated, potent inhibitory action against Galectin-3. The next crucial step involves formulating DP9 for in vivo studies to assess its pharmacokinetics, bioavailability, and efficacy in relevant animal models of pancreatic cancer. The innovative application of computational modeling alongside rigorous structural and biological assays provides a clear, actionable path toward exploiting this novel compound. DP9 represents not just a promising natural compound but a blueprint for designing a new class of Carbohydrate-based Drugs to combat highly aggressive, Gal-3-driven cancers, fueling the next wave of drug development from the ocean's unique chemical reservoir.
Reference
- Liu, P.; et al. Novel sulfated oligosaccharide DP9 from marine algae, Gracilaria lemaneiformis: A potent Galectin-3 inhibitor for pancreatic cancer therapy. Marine Drugs. 2025, 23(11): 423. (Open Access)

 Fig.1 Binding conformation of docked DP9 and Gal-3 molecules. (Liu, et al., 2025)
Fig.1 Binding conformation of docked DP9 and Gal-3 molecules. (Liu, et al., 2025) Fig.2 Mechanism of DP9 in the BxPC-3 cell targeting the Gal-3/EGFR/AKT/FOXO3 signaling pathway. (Liu, et al., 2025)
Fig.2 Mechanism of DP9 in the BxPC-3 cell targeting the Gal-3/EGFR/AKT/FOXO3 signaling pathway. (Liu, et al., 2025)



