Effects on Ulcerative Colitis of Fucoidan and Its Derivative from Saccharina japonica
November 17, 2025
On November 3, 2025, the research team of Yu, Jin, and colleagues, experts in marine natural product chemistry, published an article in the journal Marine Drugs titled " Structural Comparison and Therapeutic Effects on Ulcerative Colitis of Fucoidan and Its Derivative from Saccharina japonica". The paper investigates the structural comparison and therapeutic application of a Native Fucoidan (CF) and its degraded derivative (DF) derived from the common brown seaweed, Saccharina japonica. The core finding demonstrates that controlled degradation of this key Marine Glycan significantly reduces its molecular weight—from a complex 582 kDa polymer to a specific 2.3 kDa oligosaccharide mixture. This structural refinement significantly enhances its efficacy in treating chemically-induced ulcerative colitis (UC) in a mouse model, illuminating a promising path toward structure-activity relationship-based glycan therapy for chronic inflammatory diseases. This work elevates the marine sulfated polysaccharide, specifically fucoidan, from a general natural extract to a precision therapeutic candidate, setting a new benchmark for structural requirements in glycan drug development. The precise molecular engineering of this biopolymer is essential for unlocking its full therapeutic potential against the devastating effects of inflammatory bowel disease.
Research Background
UC is a chronic inflammatory condition of the colon that lacks universally effective treatments without significant side effects. Marine Polysaccharides, particularly CF, have long been studied for their impressive range of bioactivities, including Anti-inflammatory, Antioxidant, and immunomodulatory properties. However, their high molecular weight and structural heterogeneity in native extracts often limit their bioavailability, absorption, and clinical translatability. This research successfully addresses a critical gap by systematically comparing the activity of a complex native CF against a structurally simplified, low-molecular-weight derivative to determine the specific structural features that govern therapeutic efficacy against UC pathology.
Research Results
- Controlled Hydrolysis Yields Low-Molecular-Weight Glycan
The initial high-molecular-weight CF was successfully extracted and characterized with a calculated molecular weight of 582 kDa. The critical technical innovation lies in the subsequent controlled acid hydrolysis process, which was instrumental in creating the DF, achieving a dramatic and precise reduction in molecular weight to approximately 2.3 kDa. This shift successfully transformed a complex high-polymer glycan into a specific, lower-molecular-weight oligosaccharide mixture. Chemical analysis confirmed that while the total sulfate content remained relatively stable, the low-molecular-weight DF exhibited a much higher total sugar content (74.0%) compared to the native extract (49.6%), indicating a significant purification and concentration of the active fucose-rich glycan core.
 Fig.1 Molecular weight distribution of CF and DF. (Yu, et al., 2025)
Fig.1 Molecular weight distribution of CF and DF. (Yu, et al., 2025)
- Methylation Analysis Confirms Fucose Backbone Linkages
Glycosidic linkage analysis using advanced methylation techniques provided critical insight into the structural change. The native FC structure was confirmed to be predominantly composed of alpha-L-fucopyranose residues linked primarily through C1 and C3 (1,3-Fucp), a backbone structure characteristic of highly biologically active FC. Importantly, the degradation process resulted in a higher molar percentage of these non-reducing terminal residues in DF (62.28%) compared to CF (47.25%). Simultaneously, the percentage of more complex branching units, such as 1,3,4-Fucp, decreased substantially (14.62% to 5.82%). This structural refinement suggests that the improved Biological Activity of the derivative is directly attributable to a more linear, optimal chain length and a concentration of the potent 1,3-linked fucose backbone.
- Low-Molecular-Weight CF Mitigates Colitis Pathology
The therapeutic potential of the characterized glycans was rigorously tested in vivo using a DSS-induced murine model of UC. Both the CF and the DF provided significant clinical protection. Compared to the control model group, both treatments led to a less severe decline in body weight and markedly reduced the disease activity index (DAI) scores. Crucially, the treatments effectively reversed the characteristic colonic shortening associated with severe inflammation. Histological analysis further supported the findings, showing preserved epithelial integrity, restored crypt structure, and significantly reduced inflammatory cell infiltration in the colonic mucosa of the treated mice. This work provides strong evidence that low-molecular-weight CF derivatives are effective therapeutic candidates for managing inflammatory bowel diseases.
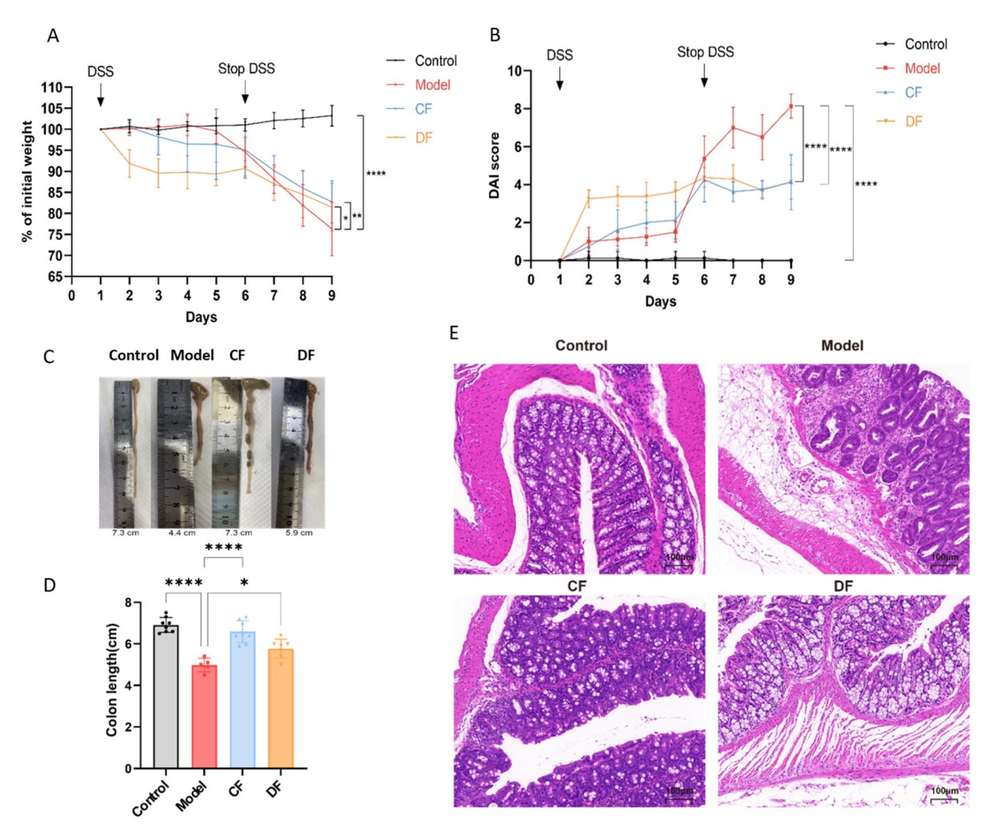 Fig.2 Effects of fucoidan treatment on basic indices of DSS-induced UC in mice. (Yu, et al., 2025)
Fig.2 Effects of fucoidan treatment on basic indices of DSS-induced UC in mice. (Yu, et al., 2025)
Conclusion
This comprehensive study provides compelling evidence that the degradation of CF from Saccharina japonica is not merely a process of simplification but rather a controlled refinement that optimizes its structural parameters for anti-colitis activity. By establishing a direct correlation between low molecular weight, specific 1,3-fucose backbone configurations, and defined sulfation patterns with enhanced therapeutic outcomes, the authors have laid a robust foundation for future glycan drug development. The innovative application of controlled hydrolysis and detailed structural analysis establishes a clear, evidence-based pathway toward developing superior functional food ingredients and novel pharmaceutical agents where precise glycan structure is the key to managing chronic inflammatory conditions like UC. Future research efforts will undoubtedly focus on synthesizing these active fuco-oligosaccharide structures chemically to achieve 100% purity and structural uniformity, moving from natural derivation to rational design. Moreover, clinical investigation into the specific mechanisms will solidify its role as a targeted therapy. This paper is a call to action for the glycobiology community to prioritize structure-activity data in the development of the next generation of highly efficacious, low-side-effect treatments derived from the vast and untapped resource of the marine environment.
Reference
- Yu, Y.; et al. Structural comparison and therapeutic effects on ulcerative colitis of fucoidan and its derivative from Saccharina japonica. Marine Drugs. 2025, 23(11): 426. (Open Access)

 Fig.1 Molecular weight distribution of CF and DF. (Yu, et al., 2025)
Fig.1 Molecular weight distribution of CF and DF. (Yu, et al., 2025) Fig.2 Effects of fucoidan treatment on basic indices of DSS-induced UC in mice. (Yu, et al., 2025)
Fig.2 Effects of fucoidan treatment on basic indices of DSS-induced UC in mice. (Yu, et al., 2025)