Vaterite/Fucoidan Hybrid Microparticles
November 10, 2025
On November 5, 2025, a collaborative team of researchers (Mosievich, Balabushevich, et al.) published an article in the journal Marine Drugs titled "Vaterite/Fucoidan Hybrid Microparticles: Fabrication, Loading of Lactoferrin, Structural Characteristics and Functional Properties." The paper details the fabrication and comprehensive Characterization of stable vaterite/fucoidan hybrid microparticles designed to encapsulate the therapeutic protein lactoferrin. The core finding is the successful integration of this key Marine Glycoconjugate to impart pH-responsive stability and controlled release kinetics, overcoming major obstacles in the non-invasive delivery of sensitive biomolecules. This work firmly establishes marine glycobiology at the forefront of advanced pharmaceutical formulation science.
Research Background
The conventional therapeutic paradigm for many high-value biologics, including peptides, proteins, and nucleic acids, is parenteral injection, largely because these molecules are prone to rapid degradation by the harsh acidic environment of the stomach and the enzymatic activity of the gastrointestinal tract. Consequently, they exhibit extremely low oral bioavailability. This work directly addresses the persistent, foundational challenge in biopharmaceutical development. By selecting and optimizing a naturally derived marine biopolymer, the research capitalizes on the intrinsic advantages of these materials—namely, their excellent biocompatibility, structural diversity, and relatively low toxicity. Positioned as a novel component, fucoidan does not merely act as a passive encapsulant; it actively provides both structural stability to the mineral phase and a biological, functional interface at the particle-tissue boundary, representing a critical leap forward from inert carrier technologies.
Research Results
The development of these novel carriers is a tour de force in nanocarrier design, requiring a deep, nuanced understanding of biopolymer-mineral interactions and glycoconjugate surface modulation. The rigorous experimental findings demonstrate exceptional control over particle characteristics, which translates directly into superior functional performance in challenging physiological environments.
- Precision Engineering of Hybrid Structures
The fabrication process relied on the co-precipitation of calcium carbonate (CaCO3) in the vaterite polymorphic form. Vaterite is thermodynamically metastable but highly desirable due to its porous structure and high specific surface area, which facilitates superior drug loading. The innovation lies in conducting this precipitation in the presence of Fucoidan, a marine fucose-rich Sulfated Polysaccharide extracted from brown algae. Fucoidan successfully functioned as a structural modifier and crystallization template. Its highly poly-anionic nature, derived from the sulfate groups, dictates the nucleation and growth of the vaterite crystals, creating stable hybrid microparticles. Critical analytical techniques, including X-ray diffraction (XRD) and scanning electron microscopy (SEM), confirmed that the fucoidan incorporation suppressed the rapid, spontaneous conversion of vaterite to the less porous and less effective calcite phase. This structural integrity is paramount, as it preserves the requisite high loading capacity for therapeutic protein delivery over extended storage and transit times, representing a substantial improvement over pure CaCO3 carriers.
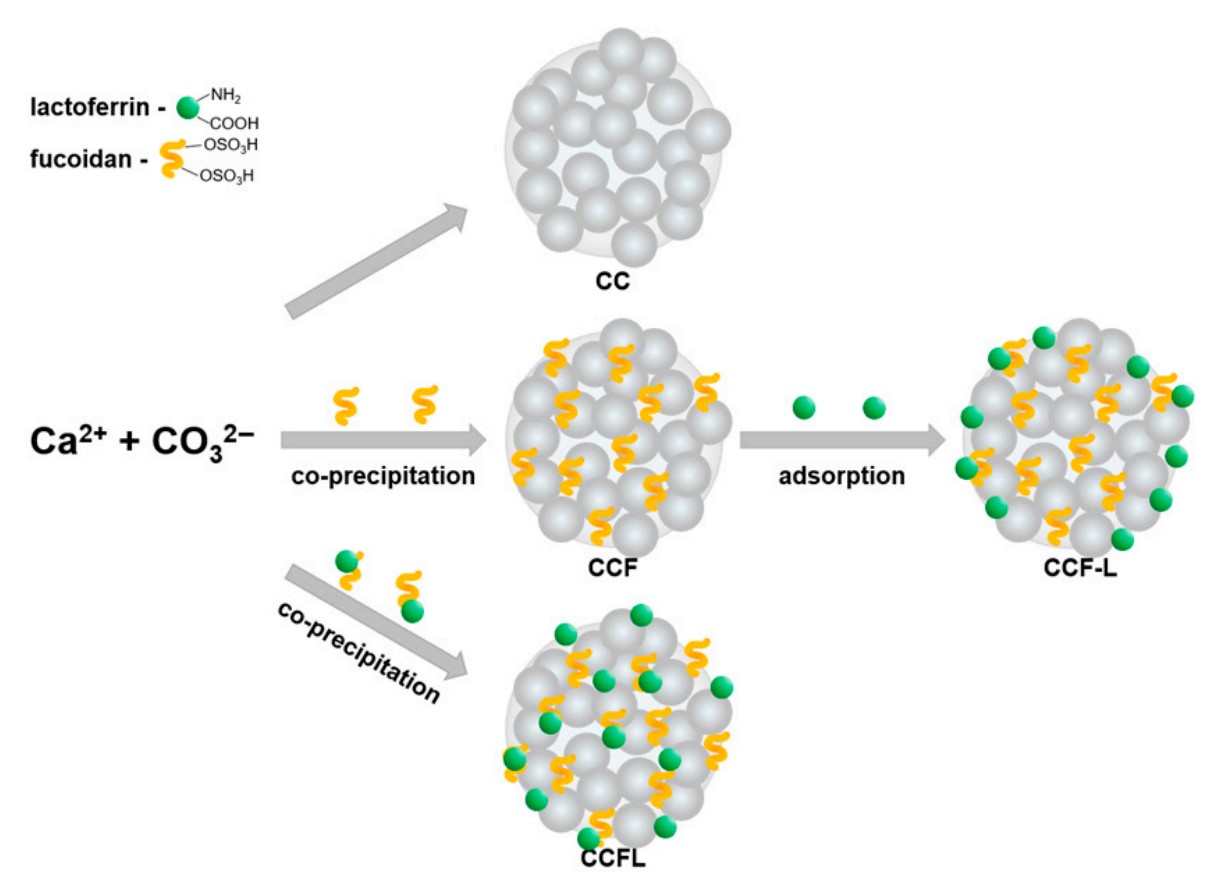 Fig.1 Scheme for obtaining control and hybrid vaterite microparticles with fucoidan and lactoferrin. (Mosievich, et al., 2025)
Fig.1 Scheme for obtaining control and hybrid vaterite microparticles with fucoidan and lactoferrin. (Mosievich, et al., 2025)
- Targeted Release and Acid Resistance Profile
Achieving resistance to the highly corrosive gastric environment is the single most critical challenge for oral delivery of protein therapeutics. The researchers meticulously evaluated the stability of the hybrid microparticles and the release profile of the loaded lactoferrin across a physiologically relevant pH gradient (ranging from pH 2-8). The results unequivocally demonstrated minimal burst release of both the polysaccharide and the protein drug under highly acidic conditions (pH 2), which accurately mimics the stomach's environment. This acid-resistant barrier function is attributed to the fucoidan shell, which acts as a protective, pH-responsive membrane. Furthermore, the kinetics analysis showed a dramatic and desirable increase in the release rate in neutral environments (pH 7.4), typical of the small intestine. This precise, "on-demand" intestinal-targeted release profile confirms the functional success of the fucoidan-based hybrid shell, validating its role as a highly effective protective and targeting system for sensitive biological payloads.
 Fig.2 Scanning electron microscopy (SEM) and diameter distribution diagram of microparticles. (Mosievich, et al., 2025)
Fig.2 Scanning electron microscopy (SEM) and diameter distribution diagram of microparticles. (Mosievich, et al., 2025)
- Glycobiological Modulation of Mucoadhesion
The biological efficacy of an oral nanocarrier is fundamentally determined by its interaction with the gastrointestinal mucosal lining, a barrier defined by the High-Molecular-Weight Glycoprotein mucin. The role of the marine glycoconjugate layer in modulating this interaction was validated through sophisticated studies utilizing fluorescently labeled mucin (FITC-mucin). Quantification of mucin adsorption onto the particle surface was conducted using fluorescence spectroscopy and flow cytometry. The hybrid particles (CCFL) exhibited significantly lower adsorption of high-molecular-weight mucin compared to bare CaCO3 microparticles. Mechanistically, this reduced interaction is hypothesized to be due to the electrostatic repulsion created by the dense, highly negative charge of fucoidan's surface-exposed sulfate groups, which effectively shields the particle from the similarly negatively charged mucin polymer chains. This level of precise control over the particle-glycoprotein interface is a profound glycobiological innovation. It suggests the potential to fine-tune microparticle transit and absorption by either promoting optimal permeation through the mucus layer or facilitating controlled residence time, thereby maximizing local drug concentration and subsequent bioavailability.
Conclusion
This research provides compelling evidence for the successful design and fabrication of a sophisticated, fucoidan-stabilized oral delivery system. The resulting glycoparticles demonstrate superior structural stability, precise pH-responsive release, and an optimized interaction profile with the mucosal barrier. The innovative integration of a functional marine glycoconjugate provides a template for significantly advancing the oral administration of numerous sensitive biological drugs. This platform holds immense promise for clinical translation, potentially replacing injectable therapeutics with patient-friendly oral formulations across various therapeutic areas.
Reference
- Mosievich, D.V.; et al. Vaterite/fucoidan hybrid microparticles: fabrication, loading of lactoferrin, structural characteristics and functional properties. Marine Drugs. 2025, 23(11): 428. (Open Access)

 Fig.1 Scheme for obtaining control and hybrid vaterite microparticles with fucoidan and lactoferrin. (Mosievich, et al., 2025)
Fig.1 Scheme for obtaining control and hybrid vaterite microparticles with fucoidan and lactoferrin. (Mosievich, et al., 2025) Fig.2 Scanning electron microscopy (SEM) and diameter distribution diagram of microparticles. (Mosievich, et al., 2025)
Fig.2 Scanning electron microscopy (SEM) and diameter distribution diagram of microparticles. (Mosievich, et al., 2025)